Histone Linker H1 Domain, Spermatid Specific 1 (HILS1)
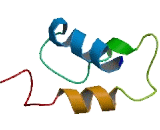
The deduced 231-amino acid protein shares 50% identity with mouse Hils1, which has a shorter N terminus. Mouse and human HILS1 each have an N-terminal domain, a globular domain, and a C-terminal domain, and both contain 2 conserved phosphorylation sites. The authors predicted that the globular domain of HILS1 forms a winged-helix structure similar to histone H1. Northern blot analysis of mouse tissues detected a 1.0-kb transcript only in testis. RT-PCR confirmed that HILS1 is expressed exclusively in testis in mouse and human. Hils1 localized to step-12 spermatid nuclei in a pattern that was similar but not identical to that displayed by Tnp1. In mouse, Hils1 displays characteristics of a linker histone and is expressed in nuclei of late maturing spermatids.
Если вы не увидели здесь нужный Вам продукт - это значит, что он доступен для изготовления на заказ.